 
Typically, the composition of a liquid injectable drug means that that a very large volume can be filtered before blocking of that filter occurs. The purpose of MFT is to provide a measure of the likelihood of microbiological contamination arising in a particular aseptic process. Sterilisation by filtering (for soluble liquids) followed by filling under aseptic conditions is the method for preparation of these types of drug. ‘Sterile for use’ liquid drugs often contain heat-sensitive components which means that terminal sterilisation by autoclaving is not an option. The placebo chosen should allow the aseptic process to be simulated exactly and the pre-sterilised TSB is added downstream of the processing 6.įor laboratory use, Oxoid standard Tryptone Soya Broth (CM0129) may be more appropriate. A decision following pre-determined guidelines can be made based on the proportion of contaminated units and the identity of the micro-organisms recovered 6.įor solid presentations where a sterile end product is required, aseptic production processes can be monitored by adding medium to a suitable placebo. The medium is then incubated and the number of contaminated units is scored versus those that are un-contaminated. Cold Filterable TSB can be used as a substitute for filter-sterilised drug products and is processed in a manner identical to that in which the product would be processed i.e. The purpose of MFT is to provide a measure of the likelihood of microbiological contamination occurring in a particular aseptic process. Packs of Cold Filterable TSB have been given a sterilising dose of gamma-irradiation (minimum 25KGy) validated as a lethal dose for all yeasts, moulds and bacteria including bacterial spores and mycoplasmas.Īqueous liquid products that are required to be sterile but cannot be terminally sterilised due to the heat-sensitive nature of one or more component, may be produced by filtering-sterilising the dissolved solution and maintaining sterility by filling and closing the product under aseptic conditions. Each component of this medium has been specially screened and selected to give a highly filterable solution. 
The formulation of Cold Filterable TSB conforms to that stated in the European Pharmacopoeia 4th Edition 2002 2, the British Pharmacopoeia 2003 3, the US Pharmacopoeia 27 NF22 2004 4 and the Japanese Pharmacopoeia XIV 2001 5. Tryptone Soya Broth is highly nutritious, general purpose medium which can support the growth of a wide range of bacteria, yeasts and moulds when incubated under the appropriate conditions 1. Refer to the appropriate regulatory body for full guidelines 2,3,4,5. Micro-organisms from any contaminated units should be sub-cultured, purified and identified to species level. Visual inspection of the units should be carried out on a daily or every second day basis. Incubation of media fills is usually carried out for 14 days 6 at both 20-25☌ and 30-35☌. The medium may be made at this concentration and then sterilised by filtration or by autoclaving at 121☌ for 15 minutes. Does not distribute, republish or otherwise provide any information or derived works to any third party in any manner or use or process information or derived works for any commercial purposes.COLD FILTERABLE TRYPTONE SOYA BROTH (TSB)Ī gamma-irradiated, cold filterable Tryptone Soya Broth suitable for microbiological Media Fill Trials (MFT) for the pharmaceutical industry.ĭownload product literature on cold filterable Tryptone Soya Broth (192KB) in PDF format.Ĭold Filterable TSB should be used at a working dilution of 30g in 1 litre of distilled water (3% w/v).Uses the information solely in relation to the management of their personal funds and not as a trader to the public or for the investment of corporate funds.Does not currently act in any capacity as an investment adviser, whether or not they have at some time been qualified to do so.Is not currently registered or qualified as a professional securities trader or investment adviser with any national or state exchange, regulatory authority, professional association or recognised professional body.Is not required to be regulated or supervised by a body concerned with the regulation or supervision of investment or financial services. 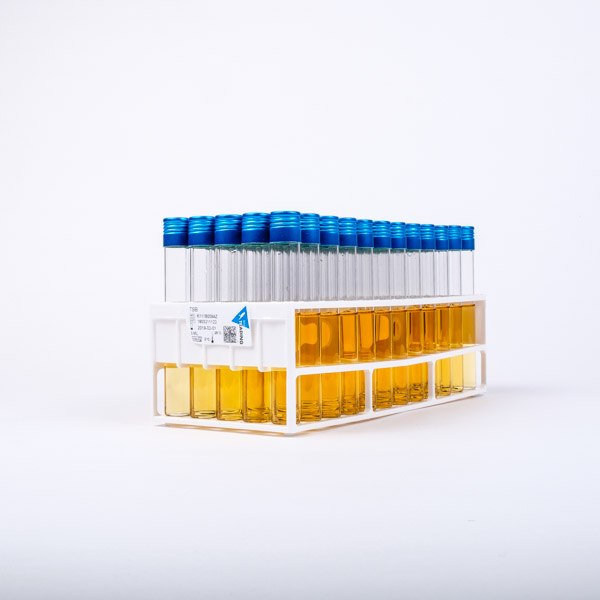
Obtains access to the information in a personal capacity.I am a private investor* I am not a private investor *A Private Investor is a recipient of the information who meets all of the conditions set out below, the recipient: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
